A beautician at the center of a troubling case involving the use of fake Botox has publicly apologized after several women experienced serious health complications as a result of receiving the unauthorized treatments. The incident, which has raised widespread concern about the risks associated with unregulated cosmetic procedures, has become a stark reminder of the potential dangers posed by counterfeit aesthetic products.
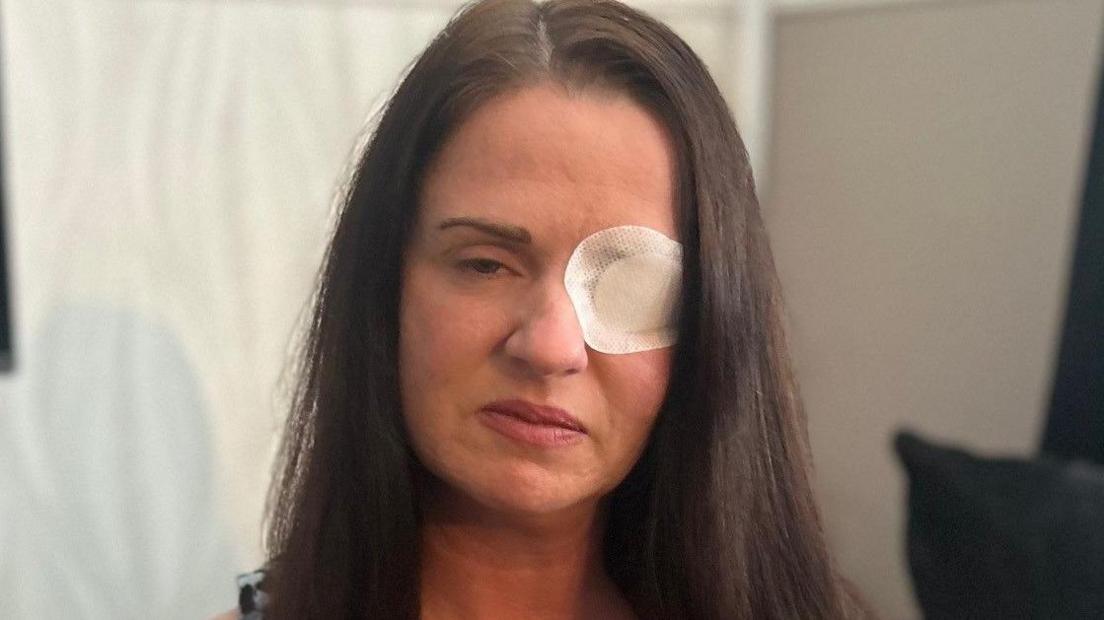
The females involved pursued Botox procedures, thinking they were obtaining authentic, FDA-endorsed injections typically employed to diminish wrinkles and fine lines. However, they were unknowingly administered with fake substances that resulted in concerning physical responses. Numerous victims experienced intense side effects, such as muscle weakness, trouble swallowing, facial paralysis, and other symptoms associated with botulinum toxin poisoning.
While accounts of the negative impacts started to emerge, health officials initiated probes to identify the origin of the fake injections. It became evident that the substances utilized were not genuine Botox produced by recognized pharmaceutical firms but were instead unauthorized, potentially smuggled in, and comprised unidentified or hazardous components.
Following the controversy, the beauty specialist who carried out the injections publicly apologized, expressing remorse for the damage inflicted and recognizing the discomfort felt by those impacted. Nonetheless, the apology has minimally alleviated the anger and anxiety among the victims and the broader community, many of whom are advocating for tighter regulations on cosmetic procedures performed outside certified medical environments.
Several of the affected women have shared their stories publicly, describing the physical and emotional toll the ordeal has taken on their lives. Some have required hospitalization, while others continue to undergo medical treatment to manage lingering symptoms. The psychological impact has also been profound, with many expressing anxiety, loss of confidence, and anger over the betrayal of trust.
Medical professionals have weighed in on the case, emphasizing the importance of ensuring that cosmetic treatments such as Botox are administered only by qualified, licensed practitioners using certified products. Unlike over-the-counter beauty treatments, injectable procedures involve substances that can carry serious risks if improperly sourced or administered. Experts stress that even when performed correctly, Botox carries potential side effects, which makes the use of counterfeit products especially hazardous.
The event has pointed out deficiencies in oversight and application within the thriving cosmetic sector. The demand for non-invasive beauty procedures has surged significantly in recent times, with numerous beauty salons, spas, and unlicensed providers offering Botox and other injectable services. While certain practitioners conduct their operations legally and ethically, others function without sufficient training or access to authentic products, endangering consumers.
Authorities have subsequently alerted the community, recommending that people check the qualifications of professionals and the authenticity of the items utilized prior to any aesthetic treatment. The U.S. Food and Drug Administration (FDA) and comparable oversight organizations in various nations have emphasized that Botox should only be provided by certified medical experts and that fake versions can pose serious health risks.
The cosmetic professional in question is currently confronting possible legal repercussions, as several impacted women are contemplating taking legal action to obtain compensation for their distress. Specialists in law indicate that the employment of fraudulent medical goods, along with the absence of appropriate licensing, might lead to considerable accountability. This situation could establish a standard for managing similar occurrences moving forward, possibly resulting in more rigorous regulations overseeing beauty services.
The demand for aesthetic enhancements continues to rise, driven by social media trends, celebrity influence, and the desire for minimally invasive solutions to aging. However, this case underscores the darker side of the industry—where shortcuts, cost-cutting, and unlicensed practices can result in harm rather than beauty.
Those affected by the counterfeit Botox treatments have faced a gradual and unpredictable recovery. Numerous individuals persist in receiving healthcare and emotional assistance as they strive to restore their self-assurance and overall health. Some have voiced optimism that by communicating their experiences, they might help avert others from encountering similar predicaments.
The general public reaction has ignited discussions regarding the factors that push people to pursue aesthetic improvements and the significance of making well-informed choices. Medical experts and advocacy organizations are urging for enhanced awareness about the dangers of non-regulated cosmetic treatments, encouraging individuals to put safety above affordability or ease.
Additionally, the cosmetic industry itself may face greater scrutiny as calls for tighter regulation grow louder. Some experts suggest that clearer guidelines, mandatory licensing, and greater transparency around the sourcing of injectable products could help protect consumers. Regulatory bodies may also increase enforcement efforts to crack down on illegal or unethical cosmetic practices.
The emotional consequences of these events should not be minimized. Those affected by unsuccessful cosmetic surgeries frequently endure both physical damage and emotional turmoil. Emotions of embarrassment, remorse, and frustration may accompany the noticeable outcomes of a procedure that has failed, highlighting the importance of psychological assistance as a crucial element of the healing process.
Moving forward, this case serves as an important cautionary tale for both consumers and practitioners. It highlights the necessity of ensuring that all cosmetic procedures—no matter how common or seemingly low-risk—are conducted within a framework of medical oversight, using products that meet strict safety standards.
The apology issued by the beautician, although a significant admission of responsibility, might fall short of mending the harm caused. Confidence in the beauty services sector is fragile, and once it’s shattered, it can be challenging to mend. For the impacted women, the physical and emotional wounds persist, highlighting the very genuine dangers associated with opting for unlicensed cosmetic procedures.
As the investigation continues and legal proceedings take shape, the case has become a wake-up call for regulatory agencies, industry professionals, and consumers alike. It is a reminder that beauty should never come at the expense of health and safety—and that vigilance, transparency, and accountability must be central to every cosmetic treatment offered.